Orientation Selective Ganglion Cell
Orientation Selective Ganglion Cells (OSGCs) are ganglion cells that respond to the alignment orientation of stimuli as opposed to the direction of movement of the stimuli. The cell-bodies of orientation selective ganglion cells are located on the vitreal side of the inner plexiform layer of the retina, however, displaced cell-bodies of ganglion cells can occasionally be found in the inner margin of the inner nuclear layer. The majority of orientation selective ganglion cells are located in the visual streak region of the retina. Orientation selective ganglion cells are categorized into ON-center and OFF-center cells. It has been observed that OFF-center orientation selective ganglion cells are more prevalent than ON-center orientation selective ganglion cells in the visual streak of the retina.[1][2]
Contents
Physiology
Receptive Field Structure
OSGCs have oval-shaped concentric receptive fields that favor either the horizontal or vertical orientation. The stimuli that were used in the study by Levick in 1967 to demonstrate the orientation selectivity of these cells were narrow strips of light oriented in different directions on the receptive fields. The cells responded to these stationary strips of light when presented at the preferred orientation of the ganglion cell. Some of these cells responded only when the strip of light was presented in the horizontal direction, and the others responded more strongly when presented in the vertical direction. It was also determined that these cells respond to strips of light when the intensity contrast between the strip of light and the background is 1/5.
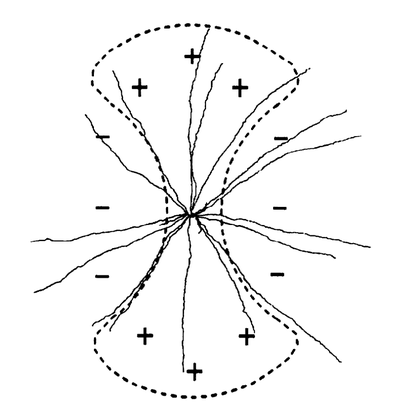
The oval-shaped receptive fields of these cells consist of a central region with an inhibitory region on either side; the central region is a strip that separates the two inhibitory regions. The illumination of both the central and two side inhibitory regions produces no response in the cell due to the cancellation of the excitatory and inhibitory responses.[1]
The center receptive field is approximately 480 micrometers in length along the axis of the preferred orientation, and approximately 230 micrometers in length along the null axis. The null inhibitory regions begin at approximately 125 micrometers from the center from either side.[3]
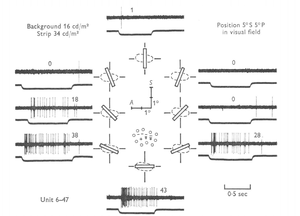
Response to Preferred Orientation
The receptive fields of these cells consist of a center, excitatory region surrounded by inhibitory sections. The intracellular electrical responses of the orientation selective ganglion cells were measured when a 95 micrometer wide/6 mm long slit of light was oriented at different angles on the receptive field. The responses recorded from both ON-center and OFF-center orientation selective ganglion cells showed that there was an initial hyperpolarization at light onset regardless of the orientation of the stimulus.
ON-center orientation selective ganglion cells have a preferred orientation that results in an initial hyperpolarization followed by a large depolarization and increased spiking when a slit of light is introduced to the receptive field of the cell. As the orientation of the slit was moved away from the preferred orientation, the initial hyperpolarization remained the same, while the depolarization decreased. At 90 degrees from the preferred orientation there was an initial hyperpolarization followed by sustained hyperpolarization.
OFF-center orientation selective ganglion cells responded with an initial hyperpolarization followed by sustained hyperpolarization and then depolarization when the light was removed from the center. When the slit of light was placed at the null angle (90 degrees from the preferred orientation) there was no longer a depolarization when the light was removed, but rather a declining hyperpolarization was observed.
It is hypothesized that the initial hyperpolarizations recorded are caused by the field peripheral to the center. The length of the slit of light was reduced, and it was found that the hyperpolarizations decreased in amplitude. When the slit of light was reduced to 300 micrometers there was an initial hyperpolarization, but the sustained hyperpolarization was replaced by depolarization. At 225 micrometers, the hyperpolarization response was no longer found and the orientation selective ganglion cells did not show a preference to orientation with this stimulus. This means that inhibition from the peripheral region of the receptive field is necessary for orientation selectivity[3]
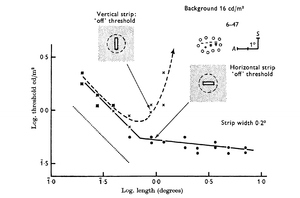
Horizontally and Vertically Selective Ganglion Cells
In a study by Venkataramani and Taylor it was observed that the orientation selectivity of vertical OSGCs is more tuned than the orientation selectivity of horizontal OSGCs. This may suggest that the spatial organization of the receptive fields of vertical and horizontal selective OSGCs are different.
Orientation selective ganglion cells receive synaptic input from amacrine and bipolar cells.[2] The responses of OSGCs may already be coded by the amacrine cells that synapse onto the ganglion cells. It is likely that these amacrine cells produce complex synaptic inputs.[3]
Orientation selectivity of these ganglion cells depends largely on GABA transmission (Caldwell, 1978). It was found that using GABA antagonists reduced the receptor's responses to the preferred orientation.[2] It has been proposed that surround inhibition is necessary to produce orientation selectivity.[1](Caldwell, 1978).
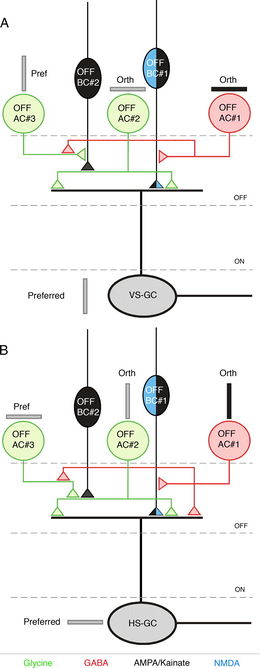
During excitation at the preferred orientation in vertically selective OSGCs, it was found that there is temporary NMDA receptor activity, sustained AMPA/kainate activity, and sustained disinhibtion (reduced glycinergic input). At the null orientation it was observed that the NMDA receptor activity and the inhbitory components were suppressed. The timing and amplitude of the excitatory and inhibitory responses recorded remained the same. This may suggest that NMDA receptor activity is not responsible for orientation selectivity.[2]
In a previous study by Caldwell, the antagonists picrotoxin and strychnine were introduced to orientation selective ganglion cells. Picrotoxin is a GABA antagonist and strychnine is a glycine antagonist; both are antagonists of inhibitory neurotransmitters. When picrotoxin was introduced to the OSGCs, the cells responded in the same way to a strip of light oriented at the preferred and null directions. With strychnine, the orientation selectivity decreased slightly(Caldwell, 1978).
Using a GABA antagonist, it was found that NMDA activity was slightly suppressed, and that the ganglion cells lost their orientation preference. The NMDA activity and disinhibition were no longer selective to orientation in vertically selective OSGCs.
At the preferred orientation in horizontal OSGCs, inhibition (GABA from amacrince cells) is reduced and AMPA/kainate and NMDA activity increases. Unlike vertical OSGCs, glycinergic disinhibition does not play a role in the excitation response, but rather there is only an increase in excitatory inputs.
OFF-center bipolar cells provide direct excitatory synaptic input to orientation selective ganglion cells with the transmission of glutamate to the NMDA and AMPA/kainate receptors. OFF-center amacrine cells inhibit the excitatory input from OFF-bipolar cells to the ganglion cells.[2]
Anatomy
Among the types of ganglion cells present in the ganglion cell layer, orientation selective ganglion cells have relatively small cell-bodies (Amthor, 1989) and have an "elongated" and "polygonal" shape (Marc, 2002).
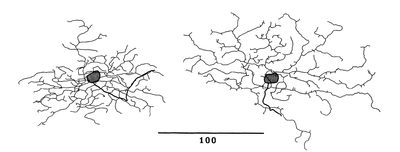
Dendritic Arbors
ON-center orientation selective ganglion cells have asymmetric dendritic arbors and have a wavy appearance (Bloomfield, 1994). The dendrites of ON-center orientation selective ganglion cells are not elongated in a particular direction that corresponds to their preferred orientation (horizontal or vertical) (Bloomfield, 1994). The dendrites of ON-center orientation ganglion cells were found to extend approximately 163 micrometers along the axis of preferred orientation (Bloomfield, 1994).
OFF-center orientation selective ganglion cells have cell-bodies that are shaped like ellipsoids and have two main dendrites extending from either side of the cell body (Bloomfield, 1994). The dendrites of OFF-center orientation selective ganglion cells are wavy in appearance, as well, and are longer than the ON-center cell dendrites (Bloomfield, 1994). The dendrites of OFF-center orientation ganglion cells were found to extend approximately 283 micrometers along the axis of preferred orientation (Bloomfield, 1994). The dendrites of both types of orientation selective ganglion cells are bistratified (Bloomfield, 1994). The extent of the dendrites of orientation selective ganglion cells has been found to be closely related to the size of the receptive field centers of OSGCs (Amthor, 1996); however a clear elongation of the dendrites along the preferred axis has not been observed.
The dendritic arbors of horizontal OSGCs are more densely branched than the dendritic arbors of vertical OSGCs.[2]
AMPA and NMDA Receptors
AMPA and NMDA receptors have been found to play a significant role in signal integration in ganglion cells found in rabbit retinas (Marc, 1999). AGB cation was used to evaluate the differences in permeability of the different ganglion cells types when kainate, AMPA, and NMDA receptors were activated (Marc, 2002).
It was found that different types of ganglion cell types respond differently to glutamate release from bipolar cells, and it is hypothesized that it may be a result of the responsivity of the different types of AMPA receptors in the ganglion cells and also the presence of GluR2 subunits (Marc, 2002).
The size of the cell and the concentration of GABA in the cell are thought to be unrelated to the responsivity of the AMPA receptors (Marc, 2002). The responsitivity of AMPA receptors in orientation selective ganglion cells is relatively high (Marc, 2002). Higher responsivity of these AMPA receptors means that signal integration time is lower for orientation selective ganglion cells (Marc, 2002).
Molecules
The neurotransmitter glycine is found in very small concentrations in ganglion cells (Marc,2002).
Compared to the other types of ganglion cells that have been identified, orientation selective ganglion cells have a relativity high GABA concentration (Marc, 2002). The neurotransmitter glutamate has also been found to be used for neural transmission by these ganglion cells, which is what differentiates these ganglion cells from amacrine cells (Marc, 2002). The molecules aspartate and glutamine have also been found to be present in orientation selective ganglion cells (Marc, 2002).
Orientation selective ganglion cells may synthesize the neurotransmitter GABA and have been found to have GABA concentrations that are very similar to those found in amacrine cells (Marc, 2002). GABA from amacrine cells may enter these ganglion cells through channels present at gap junctions(Marc, 2002). The molecule glycine is also thought to pass through these channels from amacrine to ganglion cells (Marc, 2002).
History
In 1967, Levick was the first to describe the properties of three new types of retinal ganglion cells found in the rabbit retina: orientation selective ganglion cells, local-edge detectors, and uniformity detectors.[1] Before strips of light were oriented at different angles on the receptive fields in this study, it was thought that these ganglion cells had off-center surround concentric receptive fields.[1] Levick described the receptive fields of orientation selective ganglion cells as either being horizontally or vertically selective and mentioned that the receptive fields had incomplete antagonistic surrounds.[1] He proposed that the neurons in the retina process visual information and organize information before it is sent to higher centers in the brain for further processing.[1]
In this study they found that the excitatory regions of the receptive field were difficult to find.[1]
Open Questions/Status
It is thought that amacrine cells help form the orientation selectivity of OSGCs, however, the exact role of amacrine cells still remains unclear (Bloomfield, 1994). Many questions remain on how the synaptic mechanisms create orientation selectivity in ganglion cells (Marc, 2002). A complete description of all the different types of ganglion cell types has yet to be formed and further research is needed for this to be accomplished (Marc, 2002).
Eyewire Progress
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 Levick WR (1967) Receptive fields and trigger feature of ganglion cells in the visual streak of the rabbits retina. J Physiol 188:285-307. http://jp.physoc.org/content/188/3/285.abstract?ijkey=9e2025595bfc5bfe14d04894d4c3dcd7d1b03682&keytype2=tf_ipsecsha
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Venkataramani S, WR Taylor (2010) Orientation Selectivity in Rabbit Retinal Ganglion Mediated by Presynaptic Inhibition. J Neurosci, Nov 17; 30(46):15664-15676. http://www.jneurosci.org/content/30/46/15664.full
- ↑ 3.0 3.1 3.2 3.3 Bloomfield SA(1994) Orientation-sensitive amacrine and ganglion cells in the rabbit retina. J Neurophysiol 71:1672-1691. http://jn.physiology.org/content/71/5/1672.abstract?ijkey=4cfa4066fd39257147ad90ee1df8864247a4132f&keytype2=tf_ipsecsha
- ↑ 4.0 4.1 Amthor FR, Takahashi ES, Oyster CW (1989) Morphologies of rabbit retinal ganglion cells with concentric receptive fields. Journal of Comparative Neurology 280:72-96. [1]
1. Marc RE, Jones BW (2002) Molecular phenotyping of retinal ganglion cells. J Neurosci, Jan 15;22(2):413-27. [2]
2. Marc RE (1999) Mapping glutamatergic drive in the vertebrate retina with a channel-permeant organic cation. J Comp Neurol, 407(1):47-64. [3]
5. Amthor FR, Grzywacz NM, Merwine DK (1999) Extra-receptive-field motion facilitation in on-off directionally selective ganglion cells of the rabbit retina. Visual Neuroscience 13:303-309.[4]
8. Caldwell JH, Daw NW, Wyatt HJ (1978) Effects of Picrotoxin and strychnine on rabbit retinal ganglion cells: lateral interactions for cells with more complex receptive fields. J Physiol, 276: 277-298. [5]