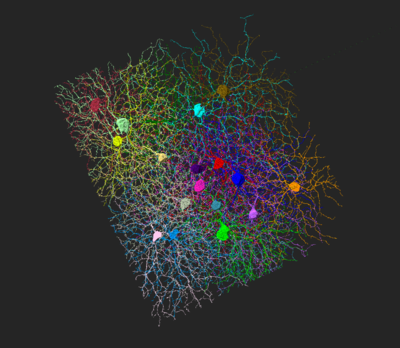
CB2-RGC
Retinal ganglion cells that express calretinin, or 29 kDa calbindin, are known as CB2-GRCs. They are a type of transient OFF-alpha RGCs (tOFF-alphaRGC). Like all alpha cells, these RGCs are immunoreactive with SMI-32, an antibody that recognizes an antigen rich in alpha RGC neurofilaments. They possess large, spherical somas, and monostratify within the OFF sublamina of the inner nuclear layer (IPL). Typical of cells that exhibit transient OFF-type physiological responses, they depolarize and spike briefly at the offset of a flash (Huberman et al., 2008). Their axons project exclusively to the superior colliculus (SC) and the dorsal lateral geniculate nucleus (dLGN).
Contents
Equivalent Classifications
- 4on: CellMuseum
- Transient OFF-alpha
Molecular Definition
These retinal ganglion cells are defined by their expression of calretinin (28 kDa calbindin). They can be visualized in calretinin-EGFP BAC transgenic mice (CB2-GFP mice), in which GFP is expressed under the calbindin 2 promoter (GENSAT). Although CB2-RGCs are the only cells in the ganglion cell layer (GCL) that express GFP in this mouse, it is also expressed by a group of amacrine cells in the inner nuclear layer (INL).
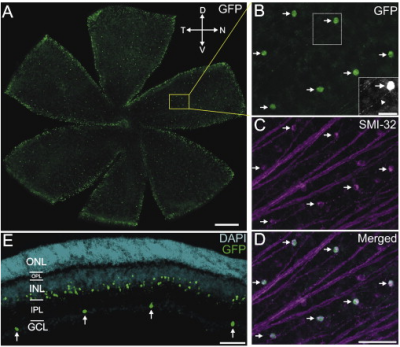
Physiology
CB2-RGCs exhibit physiological properties of transient OFF-type responses. The cells depolarize and spike at the offset of a flashing visual stimulus. (Huberman et al., 2008). Additionally, when presented with a randomly flickering visual stimulus, they show a biphasic temporal response (two distinct responses separated by time) that peaks at 105-121 ms relative to the visual stimulus (Huberman et al., 2008).
Anatomy
Dendritic Morphology
CB2-RGCs have relatively large (20-25um) and spherical cell bodies, and their dendrites stratify at the inner 30-35% depth of the IPL within the OFF sublamina (Huberman et al., 2008). They display a nonrandom mosaic across the entire retina, their cell bodies keeping a distance of 100um away from each other (Huberman et al., 2008).
Retinal Input
The exact cell types that CB2-RGCs receive input from are still unknown. However, we can infer that they have the same basic connectivity as all transient OFF-alpha type cells. Transient OFF-alpha RGCs are mediated by a transient cation current from S cone-driven bipolar cells, with additional modulation by amacrine cells with mixed rod and cone inputs (Pang et al., 2003).
Central Projections
CB2-RGCs send their axons to the contralateral superior colliculus (SC) and the dorsal lateral geniculate nucleus (dLGN). They terminate within these areas in a highly specific manner. In the SC, their axons terminate in the lower stratum griseum superficialis (lSGS), the more ventral of two layers in the SGS. Interestingly, their axons terminate in evenly spaced patches within the lSGS, forming columns of innervated and non-innervated areas (Huberman et al., 2008). This is most likely due to the fact that the projections to the SC are organized into columns of left and right-eye inputs (May, 2005, Huberman et al., 2008). In the dLGN, axons terminate within the medial dLGN (Huberman et al., 2008).
Development
The development of laminar and columnar specificity of CB2-RGC retinocollucular projections occurs between postnatal day 4 (P4) and P10 of postnatal mice (Huberman et al., 2008). Although rudimentary columns are present at P4, there is no laminar specificity; their axons innervate the entire extent of the retinorecipient SC. However, by P10, laminar and columnar organization are similar to that of the adult mouse. It was found that cholinergic retinal waves between P4-P10 mediate columnar specificity of CB2-RGCs, but are not imperative for laminar specificity (Huberman et al., 2008).
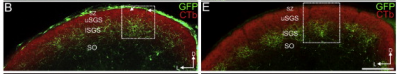
History
The use of the CB2-GFP mouse enabled central projections for a RGC type with a complete mosaic to be mapped for the first time. This cell type was discovered by Huberman et al. in 2008.
References
Huberman AD, Manu M, Koch SM, Susman MW, Lutz AB, Ulian EM, Baccus SA, Barres BA (2008). Architecture and activity-mediated refinement of axonal projections from a mosaic of genetically identified retinal ganglion Cells Neuron 59, 352-353. PubMed Free Full Text
Pang JJ, Gao F, Wu SM (2003). Light-evoked excitatory and inhibitory synaptic inputs to ON and OFF alpha ganglion cells in the mouse retina. J Neurosci 23, 6063-6073. PubMed Free Full Text
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||